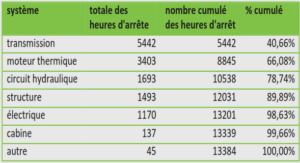
Results
GP1 interacts with an uncharacterised transmembrane protein
In order to characterize the role of GP1 in P. falciparum blood stages, we identified interacting partners by performing an anti-HA immunoprecipitation (IP) paired with mass spectrometry (MS) analysis on the previously described GP1-RFA (regulatable fluorescent affinity) tagged line (Hallee et al., 2015). The results revealed several proteins with one candidate standing out with 42 peptides identified, an uncharacterized conserved protein PF3D7_1123500 (hereafter named Golgi Protein 2, GP2). GP2 was the protein with the second highest number of peptides after GP1 (Table S1). GP2 is a transmembrane protein of 1336 amino acids conserved in Plasmodium spp. but with no significant homology outside the Plasmodium genus. A BLAST analysis (https://blast.ncbi.nlm.nih.gov/Blast.cgi) using full length GP2 failed to identify homologous proteins in related organisms like Toxoplasma and other apicomplexans (not shown). GP2 is predicted to possess a signal peptide and two transmembrane domains at its C-terminal end (Figure S1A).
GP2 potentially localises to the Golgi apparatus throughout the lifecycle of the malaria parasite
To characterize GP2 we first endogenously tagged the protein with a triple hemagglutinin (HA) epitope by single cross-over recombination. A glucosamine-inducible glmS ribozyme was also incorporated within the 3’untranslated region to allow the conditional regulation of the protein (Figure S1B) (Prommana et al., 2013). The proper integration of the 3HA-glmS cassette at the GP2 locus was verified by Southern blot (Figure S1C) in addition to PCR genotyping (Figure S1Di). Furthermore, no wild type allele was detected in the GP2-3HA- glmS tagged line after three Blasticidin drug selection cycles (Figure S1C-Dii).
We also raised GP2 specific antibodies against a synthetic peptide comprising amino acids 259 to 272. (Figure S2A). A Western Blot performed on mixed-stage parasite protein extracts shows that the anti-GP2 serum recognizes a protein at the expected size of around 150 kDa in both P. falciparum 3D7 parasites and in the GP2-3HA-glmS parasite clone. Furthermore, an anti-HA Western blot revealed a single band of the same size in the tagged line only (Figure S2B). We next localized GP2 in schizont stage parasites by immunofluorescence assay (IFA) using the anti-GP2 and the anti-HA on the GP2-3HA-glmS tagged line. Both antibodies exhibited the punctate pattern typical of a Golgi-resident protein and the important overlap demonstrated that both antibodies can specifically recognize GP2 (Figure S2C)(Struck et al., 2005, Struck et al., 2008b). The extensive colocalization between GP2 and the cis-Golgi apparatus marker Endoplasmic reticulum Retention Defective 2 (ERD2) (Elmendorf et al., 1993) confirmed that GP2 resides in the Golgi throughout the blood stages (Figure 1A). This staining pattern was conserved in the rodent malaria parasite P. berghei, where we tagged the endogenous locus of the orthologous gene PBANKA_092480 with GFP. The tagged protein was also detected in gametocytes, as well as all mosquito stage parasites including the ookinete, midgut oocysts, and sporozoites providing evidence for the constitutive expression of GP2 throughout the life cycle of the parasite (Figure 1B and Figure S3). In addition, the staining pattern of PbGP2 in the mosquito stages resembled what was previously described with PbGP1 which suggests that they might perhaps also form a complex in this phase of the life cycle (Tufet-Bayona et al., 2009).
To confirm the interaction between GP1 and GP2, we performed an anti-HA IP on GP1-RFA parasites and probed with the anti-GP2 antibody. The IP was also done on GP2-3HA-glmS and a 3D7 line as positive and negative controls, respectively. A specific 150 kDa band was seen in both tagged lines but not in the wild-type control, confirming that GP2 interacts with GP1 in P. falciparum. The absence of ERD2 in the IP eluate further suggests that GP2 does not bind in a non-specific way to all Golgi proteins (Figure 1C). An IFA on GP1-RFA late stage parasites using anti-HA and anti-GP2 antibodies showed strong co-localization between both signals which would be expected if GP1 and GP2 form a complex (Figure 1D).
Identification of a GP1 interacting domain in GP2
To characterize the GP1-GP2 interaction in more detail, in vitro pull-down assays were performed. We first decided to express two large regions situated between the SP and the first transmembrane domain of GP2 (fragments A and B) (Figure 2A). The proper expression and purification of GP2 fragments coupled to the GST tag was validated by SDS-Page and Coomassie staining (Figure S4). Incubation of the GP2 fragments with GP1-RFA parasite lysate showed that only the A construct corresponding to the region comprising amino acids 239 to 571 was able to interact with GP1 (Figure 2Bi). The A region was then further subdivided in two parts (C and D) and only the D fragment was able to pull down GP1 (Figure 2Bii). Analysis of the secondary structure of this region (corresponding to amino acids 438 to 560) using the Jpred4 platform (Drozdetskiy et al., 2015) predicted four -helices followed by one -sheet (Figure 2A). Based on this information we systematically narrowed down the interacting domain to the last two -helix and the -sheet of GP2 (AA503 to 555) (Figure 2Biii). GP1 possesses a signal peptide (SP) and no TM so it is likely found inside the Golgi (Banfield, 2011). The presence of the GP1 binding domain of GP2 between the SP and the first TM suggests that it is also inside the organelle and not facing outside, in the cytoplasm.
Identification of potential GP1 and GP2 interacting partners
To identify interactors of GP2, we performed an IP-MS analysis using the GP2-3HA-glmS line together with 3D7 parasites as negative control. Table S2 lists all the potential interacting partners with an enrichment of at least 2 fold above the control. As with the GP1-RFA IP, the proteins with the highest number of peptides were GP2 and GP1 (67 peptides and 23 peptides identified, respectively). To try to identify other proteins putatively interacting with the GP1- GP2 complex, we looked at proteins with a least a 2-fold enrichment in both IPs (Table S3). Interestingly, two proteins associated with the ERAD pathway were recovered: Ubiquitin- activating enzyme E1 (PF3D7_1225800, UBA1), and Protein disulfide isomerase (PF3D7_0827900). We also found two mitochondrial proteins, one tRNA ligase, one RNA binding protein, a protein involved in nuclear export and the enzyme 6-phosphofructokinase. Based on their known functions and subcellular localisations, these proteins are unlikely to be true interactors of the GP1-GP2 complex. Finally, the merozoite surface protein MSP1 was also identified as a potential interactor. Since MSP1 transits through the secretory pathway on its way to the parasite plasma membrane, it could indeed encounter the GP protein complex. This might suggest that the GP complex could act as an escort to MSP1 however the lack of an obvious defect in MSP1 trafficking in parasite lines where the expression of GP1 is decreased and of GP2 is completely abolished makes it unlikely (see below).
Knockdown of GP1 affects in vitro P. falciparum asexual stage parasite growth
The inability to inactivate P. berghei GP1 previously suggested that it was essential in the blood stages (Tufet-Bayona et al., 2009). To determine whether this was the case in P. falciparum, we attempted to knock out the GP1 gene using the newly developed selection- linked integration for targeted gene disruption strategy (SLI-TGD). This method provides greatly increased efficiency in the selection of parasites with genomic integrations (Birnbaum et al., 2017). Despite numerous attempts, we were unable to delete GP1 (data not shown).
We had previously unsuccessfully tried to conditionally regulate the expression of GP1 by using the DHFR destabilization domain (Muralidharan et al., 2011, Hallee et al., 2015) so we decided to switch to the glmS ribozyme system and generated a marker-free GP1-3HA-glmS tagged line by using the CRISPR-Cas9 technology (Figure S5A) (Ghorbal et al., 2014, Theriault et al., 2017). Proper integration of the 3HA-glmS cassette at the 3’end of the GP1 gene was verified by Southern Blot and PCR before and after parasite cloning (Figure S5B- C). Anti-HA Western blot on GP1-3HA-glmS protein extracts showed a band of around 80 kDa which is smaller than the expected size of 150 kDa (Figure S5D). This was also seen with the GP1-RFA strain which led us to speculate that GP1 was potentially processed. Incubation of the GP1-3HA-glmS line with glucosamine (GlcN) revealed a specific dose dependent decrease in GP1-3HA expression (Figure 3A and B). Of interest, the expression of GP2 was not affected by the down regulation of GP1 (Figure 3A). We next looked at the effect of the GP1 KD on parasite growth and saw a mild reduction in parasitaemia of around 30% after three cycles (Figure 3C). The absence of an effect of GlcN on the GP1-RFA line control demonstrates that the decreased growth observed is not due to a toxic effect of the compound (Figure 3D).
Knock out of GP2 affects GP1 levels and negatively impacts in vitro asexual stage parasite growth.
To explore the role of GP2 in the asexual erythrocytic stages we incubated the GP2-3HA- glmS line described above (Figure S1) with increasing concentrations of GlcN resulting in a dose dependent decrease in the intensity of the anti-GP2 signal compared to the anti-GAP50 loading control (Figure 4Ai). Densitometric analysis of the signal intensity showed an almost complete knockdown of the GP2-3HA protein at 2.5 mM GlcN (Figure 4Aii). We next performed growth curve analysis in the presence or absence of 2.5 mM GlcN and the parasitaemia was monitored every 24 h for 120 h (three life cycles) by FACS (Figure 4B). Despite the almost complete absence of GP2-HA, no effect was seen on parasite growth when compared to a wild type control suggesting that either GP2 is not essential for asexual life cycle progression, or that minimal protein levels are sufficient to perform its function. To try to confirm this, we attempted to inactivate the GP2 gene using SLI-TGD (Figure S6A). In contrast to GP1, we succeeded in knocking out the GP2 gene which was confirmed by PCR detection of the proper integration of the pSLI-TGD-GP2 vector at the GP2 locus and the absence of the WT allele (Figure S6B). A Western blot using the anti-GP2 antibody showed that the GP2 protein was no longer present in the GP2KO line (Figure S6C). Analysis of the growth of the GP2KO strain showed a decrease of around 25% in the parasitemia compared to the 3D7 control (Figure S6D) demonstrating that GP2 is not essential but still plays an important role in in vitro asexual growth.
To test the effect of a simultaneous decrease in both GP1 and GP2 protein expression we tried to knock out GP2 in the GP1-3HA-glmS tagged line using SLI-TGD (Figure S7A). Proper integration of the vector and disappearance of the WT allele were detected by PCR (Figure S7B) whilst the absence of detectable GP2 by Western blot using the anti-GP2 antibody confirmed that we had successfully inactivated the GP2 gene in the GP1-3HA-glmS line (Figure S7C). Intriguingly, inactivating GP2 led to a decrease of around 75% in the amount of GP1-3HA to even lower levels than in the GP1-3HA-glmS knocked down line (i.e. incubated with 2.5 mM GlcN, around 50% decrease in GP1-3HA levels) (Figure 5A and B). Knocking down GP1-3HA in the GP2KO/GP1-3HA-glmS line led to a further reduction in its expression (around 90% reduction compared to GP13HA-glmS levels) (Figure 5A and B). Some reduction in the level of GP1-3HA was also observable by IFA though it did not look as extensive as on the Western blots (Figure 5Ci vs iii and iv). In addition, the usual punctate fluorescence pattern of GP1-3HA was slightly disrupted in the GP2 KO with some small portion of it looking like it remained trapped close to the nucleus, potentially in the ER (Figure 5Ci vs iii). By increasing the exposure time to capture more HA signal, we saw that the residual punctate pattern still colocalised with ERD2 suggesting that in absence of GP2, a significant amount of GP1-3HA was still trafficked to the Golgi (Figure 5D). Interestingly, this sort of phenotype is reminiscent, though certainly not as striking, of what was seen with the low molecular weight rhoptry protein complex where RAP1 escorts RAP2 to the rhoptries and in absence of the former, the latter stays trapped in the ER. Though a decrease in the levels of RAP2 in the RAP1KO strain is seen by Western, it is not that striking when looking at IFA images (Baldi et al., 2000). Globally, these results suggest that GP2 might potentially be required for the optimal localization of GP1 to the Golgi apparatus.
To determine the effect of the absence of GP2 combined with the reduced expression of GP1- 3HA on parasite survival, we followed the growth of the parasites over three cycles by FACS. As seen in the GP2KO in the 3D7 WT line, inactivating GP2 in the GP1-3HA-glmS line led to a 25% reduction in growth (Figure 5E: dark blue curve, KO-GP2-GP1-glmS-0mM vs dark green curve, GP1-3HA-glmS 0mM), around what we obtained with the GP1 KD line (Figure 5E: light green curve, GP1-3HA-glmS-2.5mM vs dark green curve, GP1-3HA-glmS 0mM). Decreasing the expression of GP1-3HA in the GP2 KO line led to an additive effect with a 44% reduction in parasitaemia (Figure 5B: light blue curve, KOGP2-GP1-glmS- 2.5mM vs dark green curve, GP1-3HA-glmS 0mM). Globally, these data suggest that the GP1-GP2 protein complex is important but perhaps not essential for the in vitro erythrocytic cycle of P. falciparum.
In order to provide insight into the function of the GP1-GP2 complex, we further characterized the GP1-3HAKD/GP2KO parasite line. We first followed the evolution in stage distribution of the parasites over three growth cycles and did not see any changes (Figure S8A). Next, analysis of the invasiveness of purified merozoites showed no deficiency (Figure S8B). Finally, we investigated by IFA whether the localisation of different organellar markers was affected in the KD/KO line. Only some minor effects were noticeable for the microneme markers AMA1 and EBA175, the inner membrane complex protein GAP45 and the merozoite surface protein MSP1 (Figure S9). Globally, the lack of any obvious specific defect might suggest that general parasite fitness is affected.
Inactivation of P. berghei GP2 reveals that it is dispensable throughout the life cycle
To investigate the role of GP2 in the virulence to mice and on the mosquito stage, we inactivated the GP2 gene in P. berghei (Figure 6A). The rodent malaria gene deletion mutant infected the mosquito vector similar to WT numbers. Infectious sporozoites isolated from salivary glands showed no apparent defects in motility (Figure 6B and C) and produced patent mouse infections between days 4 and 5 (Figure 6D), comparable to the WT strain. This demonstrates that PbGP2 is not required at any step of the rodent malaria life cycle.
In conclusion, we have identified a Plasmodium-specific Golgi protein complex important but likely not essential for the in vitro asexual erythrocytic cycle of the human parasite P. falciparum. Moreover, we show that P. berghei GP2 is not required for the mosquito stages nor for virulence in a mouse model of infection.