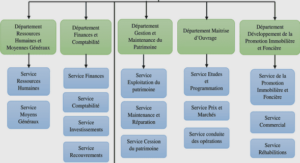
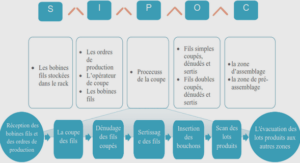
Hand specimens
Disseminated, matrix and breccia assemblages
Disseminated sulfide sample consist mainly of a troctolite with uniform texture and containing minor fine (0.2 mm) patches of sulfide. About 95 % of the rock consists of troctolite and 5 % consist of sulfides. In the matrix sulfide , sulfides form the matrix to troctolite patches (2-5 mm). The breccia sulfide contains patches (3-4 cm) of sulfides and the matrix of the rock is a troctolite. For matrix and breccia samples, the sulfide abundance correspond of ~ 45 %, the silicate consists of ~ 45 % and magnetite ~ 10% of the sample.
Massive sulfides – Fe-rich and Cu-rich
The massive sulfides samples can be divided into Fe-rich and Cu-rich assemblages. Pyrrhotite is the main sulfide mineral in the Fe-rich assemblage and forms the matrix to the other minerals. Coarse grains of pentlandite (2-3 cm) and chalcopyrite (1-1.5 cm) occur in association with pyrrhotite . Magnetite grains are generally 0.2 to 0.3 mm, but can be larger (0.7 mm – 1cm) in some Fe-rich massive sulfide samples . For Fe-rich samples ~ 60 % of the rock consists of pyrrhotite, ~ 20 % of pentlandite, ~ 10% chalcopyrite and ~ 10 % magnetite.
Chalcopyrite and Cubanite represent the main sulfide minerals in the Cu-rich assemblage, and occur as the matrix in Cu-rich samples. Coarse grains of pentlandite (1 – 2.5 cm) and pyrrhotite (1 – 3 cm) are also present. Medium grains of magnetite (0.5 cm) occur as inclusions in the chalcopyrite / cubanite matrix , but can be associated with pyrrhotite, or in contact with pentlandite. Figure 6 D shows a Cu-rich sample, where cubanite / chalcopyrite represent the matrix, containing few fine grains of magnetite (0.1 cm) and medium grains of pentlandite and pyrrhotite (0.5 – 1 cm). For VB6, ~ 50 % consists of cubanite / chalcopyrite, ~ 25 % of magnetite, ~ 15% of pentlandite and ~ 10 % of pyrrhotite. For VB34 the mineral abundance consists of ~ 70 % of cubanite / chalcopyrite, ~ 15 % of pentlandite, ~ 10 % pyrrhotite and ~ 5 % of magnetite.
Polished section descriptions
Naldrett et al. (2000b) and Huminicki (2007) have previously described the mineralogy and textures of sulfide minerals at the Ovoid ore body, and Li and Naldrett (1999) and Naldrett and Li (2007) have previously described in detail all rock types that comprise the Voisey’s Bay intrusion.
Disseminated sulfide
In VB2, pyrrhotite is the most common sulfide mineral, it has an anhedral form and the grains range from 0.05 – 0.8 mm. Pyrrhotite is in contact with pentlandite, chalcopyrite and silicates . Rare granular pentlandite and few chalcopyrite grains occur and their grains size range from 0.040 – 0.100 mm, they also have an anhedral and subhedral form. They are observed in contact with pyrrhotite and silicate minerals . Mineral abundance corresponds of ~ 95 % silicates and ~ 5 % of sulfides (predominantly pyrrhotite) .
Breccia sulfide
• Pyrrhotite and troilite
In VB 8, pyrrhotite is the predominant sulfide and it represents the matrix to the other sulfides. It has a grain size up to 1 cm and is anhedral in form. Pyrrhotite is observed in contact with pentlandite, chalcopyrite, magnetite and ilmenite. It contains exsolutions lamellae of troilite . Troilite exsolution has previously been reported by Naldrett et al. (2000b) and Huminicki (2007).
• Pentlandite
Granular pentlandite occurs as anhedral and subhedral forms, ranging from 0.1 – 0.9 mm. Pentlandite is observed mainly in contact with pyrrhotite, chalcopyrite, silicate minerals and in rare cases with magnetite / ilmenite . Chalcopyrite, silicate minerals and minor magnetite occurs as inclusions and cracks are filled with silicate minerals.
• Chalcopyrite
Chalcopyrite has an anhedral form and ranges from 0.1 – 1.4 mm. It contains silicate mineral inclusions. Chalcopyrite is in contact mainly with pyrrhotite, pentlandite and in some cases with magnetite .
• Magnetite and ilmenite
Magnetite has an anhedral (rounded or elongate shape) to subhedral form with grain size ranging from 0.05 – 1.4 mm. Magnetite occurs in textural equilibrium with ilmenite, pyrrhotite, chalcopyrite and pentlandite . Ilmenite has a similar shape and size to magnetite and is usually associated with the magnetite. In many cases ilmenite occurs in contact with magnetite and in some cases wraps around the edges of magnetite .
1. CHAPTER 1 – INTRODUCTION |